jimbosmith316
MuscleChemistry
Last week's "On Very Short Notice" item on the ability of TUDCA, i.e. tauroursodeoxycholic acid to stimulate the conversion of the "inactive" thyroid hormone T4 to its active counterpart T3 has caused quite a stir and instigated questions related to appropriate dosages and other potential benefits of a previously largely overlooked bile acid. With the recent interest from "using" muscle heads looking to protect their liver and overall health, however, the demand for TUDCA appears to be so high that whichever source Nutraplanet, the only major retailer I know of that has been selling bulk TUDCA at a reasonable price, must have gotten overwhelmed. This and Fatfree's personal request are reason enough for me to compile a brief write-up that may provide you with some clues on whether or not it may be worth to look for alternative sources on the web or even start your own little TUDCA import/export business.
What is tauroursodeoxycholic acid aka TUDCA ?
Tauroursodeoxycholic acid is a bile acid also known as TUDCA formed in the liver by conjugation of deoxycholate with taurine, usually as the sodium salt. While Western medicine has only gotten wind of the anti-apoptotic effects and its ability to protect mitochondria from cellular elements that would otherwise interfere with energy production within the past 20 years, or so, bear bile - you guessed it, a natural source of TUDCA - has an over 3,000 yearlong history of being used to treat visual disorders (Boatright. 2006), if which we know today that many of them can be treated and prevented by the administration of TUDCA and other bile-acids or bile-acid precursors, such as taurine (click here to read all about taurine)
So what's the suggested dose? That's actually quite a tricky question, because the number of human studies can be counted on the fingers of one hand (and this is still an exaggeration) and the available rodent data is mostly based on studies where the TUDCA was injected into the peritoneal cavity, which - depending on the compound that is used - will usually yield a bioavailabilty that's higher than what you will see with oral ingestion of the compound, yet lower than from intravenous injectionsAll of the following values are calculated for an 80kg human using the standard HED formula and are based on rodent studies showing benefits for the respective organ:Heart - 650mgDiabetes - 975mgLiver - 400-3,200mgAlzheimer's - 4,000mg*Parkinson's - 350mgPancreas - 3,250mg* This is the only value that's based on oral administration for the others I simply assumed 1:1 bioavailability which is probably already an overestimation for the liver and certainly for other tissues, since part of the TUDCA probably won't survive the first pass through the liver.
The distinction between different bile acids is difficile and beyond the scope of this mini-overview. What all of them have in common is that they are physiological detergents that facilitate excretion, absorption, and transport of fats and sterols in the intestine and liver. Bile acids are also steroidal amphipathic molecules derived from the catabolism of cholesterol, which makes them the ideal partner for both "water" and "fat soluble" molecules and allows them to do their job as major modulators of lipid secretion, bile flow and the absorption of dietary fats and vitamins. Aside from these long-established functions their role as regulators of key enzymes involved not only in cholesterol homeostasis, but also whole energy homeostasis has attracted more and more interest over the last years.
The unique detergent properties of bile acids are essential for the digestion and intestinal absorption of hydrophobic nutrients.No effect without "side effect"! Yet despite their metabolic utility bile acids have potent toxic properties and can - at high dosages - actually disrupt the very membranes they help to protext when they are administered at lower doses, therefore their accumulation in the blood and tissue is usually tightly regulated. And while Martinez-Diez et al. have shown that conjugated.bile acids have a very high toxicity threshold compared to their unconjugated cousins (Martinez-Diez. 2000), we are most probably still dealing with one of those classic U-shaped dose-response curves for TUDCA (with no effects at very low, beneficial effects at medium levels and toxicity issues at high levels) which are so ubiquitous in nature and still so difficult to understand for someone acculturated to the typical Western "more is more" mentality (Zinc would be another example, by the way; cf. "Zinc: 15mg is Plenty!").
A non-exhaustive list of proven benefitsWith its modulatory effects on the cell cylce regulator c-Jun N-terminal kinase (JNK), the generation and scavenging of radical oxigen specimen and glutathione S-transferase (GST) activity, which is necessary for the masteroxidant glutathione to do its job, TUDCA effectively prevented / reduced neurodegenation in rodent model of Parkinson's disease (Castro-Caldas. 2012). It has also been shown to reduce the amyloid beta-induced synaptic toxicity that is so characteristic of Alzheimer's disease (Nunes. 2012; Ramalho. 2012)Similar to taurine TUDCA appears to play an important role in cell osmolity, and has been shown to inhibit endoplasmic reticulum stress (ER) in embryonic development and ventricular contractile dsyfunction (malfunction of the heart) due to type II diabetes, fatty acid accumulation in the heart (Hua. 2010) and preserve cone density in the eye (Kim. 2012; Takada. 2012; Zhang. 2012a,b). The same anti-ER mechanism also protects the insulin releasing pancreatic beta cells of Wistar rats (Lee. 2010; Tang. 2012) and the kidney (Gao. 2012) from damage due to increased blood glucose. And Rivard et al. were even able to show that 400mg/kg of TUDCA, when they were administered intravenously were able to reduce apoptosis (death of hear cells) following myocardial infarction in rats (Rivard. 2007).TUDCA has also been shown to ameliorat insulin resistance in hypertrophic adipocytes (fat cells that burst from the seams; Jiao. 2011, Yoshizaki. 2012), to keep the expression of the adiponectine up (Zhou. 2010) and the inflammatory induced neovascularization in type II diabetes in check (Amin. 2012). Despite the fact that TUDCA is also effective against endoplasmic rectilium stress in the liver and in skeletal muscle it cannot save a methionine and choline deficient liver from getting clogged with fat (Henkel. 2012), or a palmitate (a saturated fatty acid) treated myotube from becoming insulin resistant, (Rieusse. 2012). What it can do, however is protect liver from apoptosis induced by natural or synthetic PPAR-gamma ligands (see "TTA + Fish Oil - Fat Burning Superfats or Hepatoxic Pro-Oxidants?" and "TTA + Fish Oil Revisited - Increased Intramuscular Omega-3 Levels Compromise Heart and Skeletal Muscle Performance"; cf. Nonaka. 2008) and ethanol-feeding (Colell. 2011)Interestingly the same anti-ER effects that protect the heart, the pancreas, the kidneys and, in the presence of adequate choline and methionine, also the liver, will also inhibit influenza A viral replication (Hassan. 2012; in-vitro data)TUDCA has also been found to have antibacterial effects, or rather to prevent the cytotoxic effects of Clostridium sordellii lethal toxin (CSLT) from virulent strains of Clostridium sordellii (Schulz. 2009) Only when it's conjugated to taurine UDCA (then T + UDCA = TUDCA ;-) will promote hydrocholeresis or, put simply, the output of bile acid (Úriz. 2011; intravenous administration). In view of the emerging importance of bile acids in overall energy expenditure and thyroid function (Ockenga. 2012).
This observation could well-explain the effects TUDCA has on the conversion of T4 => T3, which spiked your interest in the last installment of "On Short Notice", here at the SuppVersity (see "On Short Notice July 21, 2012").In this context it may also be wort mentioning that Nathanson et al. observed a direct stimulative effect of UDCA, the taurine devoid cousin of TUDCA on hepatic ATP secretion (Nathanson. 2001) and Drack et al. report that subcutaneous injections of 500 mg/kg in 0.15 M NaHCO3 to obesity prone mice reduced their weight gain by -22%; a significant effects on "normal" mice could was yet not observed in the study (Drack. 2012). And what about humans? As mentioned earlier, the number of studies in which TUDCA was actually administered to human beings is negligible and of actual relevance in the current context are probably on these two:Improved liver & muscle, but not adipose tissue insulin sensitivity in obese men and women (Kars. 2010) The main finding of this randomized double-blind study in which 20 obese subjects ([means +/- SD] aged 48 +/- 11 years, BMI 37 kg/m²) were assigned to receive either TUDCA at a dose of 1,750 mg/day or a placebo was a highly significant increase in insulin sensitivity (~30%; p < 0.05), which was - and this is would actually not be a bad thing for a physical culturist - liver and muscle specific!In combination with it's effect on the muscular and hepatic expression of p-AKT, this could make TUDCA the nutrient repartitioner (R-)ALA is not (cf. "Lean & Muscular with Alpha Lipoic Acid?"), if those effects would translate to individuals with normal insulin sensitivity (they do translate into human muscle cells in the Petri dish, which are protected against glucosamine induced decreases in GLUT-4 activity, when enough TUDCA was present in the incubation media; cf. Raciti. 2010).Surprisingly ineffective adjuvant after liver transplantation (Angelico. 1999) While you would expect that something that is good for liver health would also help as an adjuvant to the treatment of patients who received a liver transplant, the 16 subjects who were randomized to the active 2x250mg TUDCA treatment arm of the study did not see any statistically significant benefit in terms of one-year actuarial survival.Although the latter of these studies may actually be somewhat disappointing, its results are actually quite educative - after all, they put another emphasis on something I have touched upon in numerous previous blogposts, already: Even in cases as the one at hand, where the presence of statistically reduced serum cholesterol levels (p < 0.02) and less exuberant cholestasis confirm that a certain compound, in this case TUDCA, worked in vivo similarly to what it did in vitro, this does by no means implicate that it will also yield the real world results you would expect.

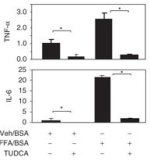
Figure 1: Anti-inflammatory effect of TUDCA on FFA treated adipocytes; reduction in TNF-alpha and IL-6 (also vs. baseline; directly from Jia. 2011))

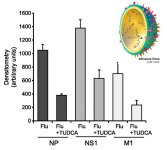
Figure 2: Western blot analysis of influenza NP, NS1, and M1 proteins with corresponding densitometry displayed as averages with S.E. Proteins - reduced expression indicate a reduced replication rate (adapted from Hassan. 2011)

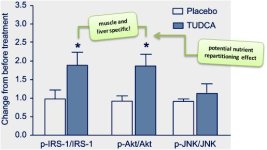
Figure 3: Effect of placebo or TUDCA on skeletal muscle insulin receptor substrate (IRS), Akt and JNK levels (Kars. 2010)
Implications: The "negative" or rather NULL outcome of the study by Angelico et al. should have reminded you that you better display a healthy degree of skepticism towards all the previously presented "scientifically established" benefits of TUDCA. Neither I, nor any of the researchers (not even Kars et al.) should have the hubris to tell you that taking X amounts of tauroursodeoxycholic acid will save your liver from the toxic effects of oral anabolic steroids, will help you in your battle against diabesity or make a powerful ally in your quest for a leaner, more muscular physique. And still, this mini-review of the literature should have shown you that TUDCA has a lot of potential, future studies will yet have to confirm, for whom these potentials can actually be realized and what dosages of a hitherto pretty pricey supplement will be necessary to produce the desired results... Needless to say that the SuppVersity is going to be the place wheer you will read about these studies first- right?
References: Amin A, Choi SK, Galan M, Kassan M, Partyka M, Kadowitz P, Henrion D, Trebak M, Belmadani S, Matrougui K. Chronic inhibition of endoplasmic reticulum stress and inflammation prevents ischaemia-induced vascular pathology in type II diabetic mice. J Pathol. 2012 Jun;227(2):165-74. Boatright JH, Moring AG, McElroy C, Phillips MJ, Do VT, Chang B, Hawes NL, Boyd AP, Sidney SS, Stewart RE, Minear SC, Chaudhury R, Ciavatta VT, Rodrigues CM, Steer CJ, Nickerson JM, Pardue MT. Tool from ancient pharmacopoeia prevents vision loss. Mol Vis. 2006 Dec 29;12:1706-14. Burroughs AK, Westaby D: Liver, biliary tract and pancreatic disease in Clinical Medicine, Eds. Kumar P, Clark M. Elsevier 2002Castro-Caldas M, Carvalho AN, Rodrigues E, Henderson CJ, Wolf CR, Rodrigues CM, Gama MJ. Tauroursodeoxycholic Acid Prevents MPTP-Induced Dopaminergic Cell Death in a Mouse Model of Parkinson's Disease. Mol Neurobiol. 2012 Jul 8.Chen Y, Liu CP, Xu KF, Mao XD, Lu YB, Fang L, Yang JW, Liu C. Effect of taurine-conjugated ursodeoxycholic acid on endoplasmic reticulum stress and apoptosis induced by advanced glycation end products in cultured mouse podocytes. Am J Nephrol. 2008;28(6):1014-22. Epub 2008 Jul 23.Colell A, Coll O, García-Ruiz C, París R, Tiribelli C, Kaplowitz N, Fernández-Checa JC. Tauroursodeoxycholic acid protects hepatocytes from ethanol-fed rats against tumor necrosis factor-induced cell death by replenishing mitochondrial glutathione. Hepatology. 2001 Nov;34(5):964-71.Drack AV, Dumitrescu AV, Bhattarai S, Gratie D, Stone EM, Mullins R, Sheffield VC. TUDCA slows retinal degeneration in two different mouse models of retinitis pigmentosa and prevents obesity in Bardet-Biedl syndrome type 1 mice. Invest Ophthalmol Vis Sci. 2012 Jan 5;53(1):100-6.Gao X, Fu L, Xiao M, Xu C, Sun L, Zhang T, Zheng F, Mei C. The nephroprotective effect of tauroursodeoxycholic Acid on ischaemia/reperfusion-induced acute kidney injury by inhibiting endoplasmic reticulum stress. Basic Clin Pharmacol Toxicol. 2012 Jul;111(1):14-23.Hassan IH, Zhang MS, Powers LS, Shao JQ, Baltrusaitis J, Rutkowski DT, Legge K, Monick MM. Influenza A viral replication is blocked by inhibition of the inositol-requiring enzyme 1 (IRE1) stress pathway. J Biol Chem. 2012 Feb 10;287(7):4679-89. Epub 2011 Dec 22.Henkel AS, Dewey AM, Anderson KA, Olivares S, Green RM. Reducing endoplasmic reticulum stress does not improve steatohepatitis in mice fed a methionine- and choline-deficient diet. Am J Physiol Gastrointest Liver Physiol. 2012 Jul;303(1):G54-9. Hua Y, Kandadi MR, Zhu M, Ren J, Sreejayan N. Tauroursodeoxycholic acid attenuates lipid accumulation in endoplasmic reticulum-stressed macrophages. J Cardiovasc Pharmacol. 2010 Jan;55(1):49-55. Jiao P, Ma J, Feng B, Zhang H, Diehl JA, Chin YE, Yan W, Xu H. FFA-induced adipocyte inflammation and insulin resistance: involvement of ER stress and IKKβ pathways. Obesity (Silver Spring). 2011 Mar;19(3):483-91. Lee YY, Hong SH, Lee YJ, Chung SS, Jung HS, Park SG, Park KS. Tauroursodeoxycholate (TUDCA), chemical chaperone, enhances function of islets by reducing ER stress. Biochem Biophys Res Commun. 2010 Jul 9;397(4):735-9.Kars M, Yang L, Gregor MF, Mohammed BS, Pietka TA, Finck BN, Patterson BW, Horton JD, Mittendorfer B, Hotamisligil GS, Klein S. Tauroursodeoxycholic Acid may improve liver and muscle but not adipose tissue insulin sensitivity in obese men and women. Diabetes. 2010 Aug;59(8):1899-905.Kim JS, Song BS, Lee KS, Kim DH, Kim SU, Choo YK, Chang KT, Koo DB. Tauroursodeoxycholic Acid Enhances the Pre-Implantation Embryo Development by Reducing Apoptosis in Pigs. Reprod Domest Anim. 2011 Dec 13.Martinez-Diez MC, Serrano MA, Monte MJ, Marin JJ. Comparison of the effects of bile acids on cell viability and DNA synthesis by rat hepatocytes in primary culture. Biochim Biophys Acta. 2000 Feb 21;1500(2):153-60.Nathanson MH, Burgstahler AD, Masyuk A, Larusso NF. Stimulation of ATP secretion in the liver by therapeutic bile acids. Biochem J. 2001 Aug 15;358(Pt 1):1-5.Nonaka M, Tazuma S, Hyogo H, Kanno K, Chayama K. Cytoprotective effect of tauroursodeoxycholate on hepatocyte apoptosis induced by peroxisome proliferator-activated receptor gamma ligand. J Gastroenterol Hepatol. 2008 Jul;23(7 Pt 2):e198-206.Nunes AF, Amaral JD, Lo AC, Fonseca MB, Viana RJ, Callaerts-Vegh Z, D'Hooge R, Rodrigues CM. TUDCA, a Bile Acid, Attenuates Amyloid Precursor Protein Processing and Amyloid-β Deposition in APP/PS1 Mice. Mol Neurobiol. 2012 Jun;45(3):440-54. Ockenga J, Valentini L, Schuetz T, Wohlgemuth F, Glaeser S, Omar A, Kasim E, duPlessis D, Featherstone K, Davis JR, Tietge UJ, Kroencke T, Biebermann H, Köhrle J, Brabant G. Plasma bile acids are associated with energy expenditure and thyroid function in humans. J Clin Endocrinol Metab. 2012 Feb;97(2):535-42.Ramalho RM, Nunes AF, Dias RB, Amaral JD, Lo AC, D'Hooge R, Sebastião AM, Rodrigues CM. Tauroursodeoxycholic acid suppresses amyloid β-induced synaptic toxicity in vitro and in APP/PS1 mice. Neurobiol Aging. 2012 May 21.Rieusset J, Chauvin MA, Durand A, Bravard A, Laugerette F, Michalski MC, Vidal H. Reduction of endoplasmic reticulum stress using chemical chaperones or Grp78 overexpression does not protect muscle cells from palmitate-induced insulin resistance. Biochem Biophys Res Commun. 2012 Jan 6;417(1):439-45. Sokol RJ, Dahl R, Devereaux MW, Yerushalmi B, Kobak GE, Gumpricht E. Human hepatic mitochondria generate reactive oxygen species and undergo the permeability transition in response to hydrophobic bile acids. J Pediatr Gastroenterol Nutr. 2005 Aug;41(2):235-43.Takada A, Miki T, Kuno A, Kouzu H, Sunaga D, Itoh T, Tanno M, Yano T, Sato T, Ishikawa S, Miura T. Role of ER Stress in Ventricular Contractile Dysfunction in Type 2 Diabetes. PLoS One. 2012;7(6):e39893. Epub 2012 Jun 29.Tang C, Koulajian K, Schuiki I, Zhang L, Desai T, Ivovic A, Wang P, Robson-Doucette C, Wheeler MB, Minassian B, Volchuk A, Giacca A. Glucose-induced beta cell dysfunction in vivo in rats: link between oxidative stress and endoplasmic reticulum stress. Diabetologia. 2012 May;55(5):1366-79.Úriz M, Sáez E, Prieto J, Medina JF, Banales JM. Ursodeoxycholic acid is conjugated with taurine to promote secretin-stimulated biliary hydrocholeresis in the normal rat. PLoS One. 2011;6(12):e28717. Epub 2011 Dec 14. Raciti GA, Iadicicco C, Ulianich L, Vind BF, Gaster M, Andreozzi F, Longo M, Teperino R, Ungaro P, Di Jeso B, Formisano P, Beguinot F, Miele C. Glucosamine-induced endoplasmic reticulum stress affects GLUT4 expression via activating transcription factor 6 in rat and human skeletal muscle cells.Diabetologia. 2010 May;53(5):955-65. Rivard AL, Steer CJ, Kren BT, Rodrigues CM, Castro RE, Bianco RW, Low WC. Administration of tauroursodeoxycholic acid (TUDCA) reduces apoptosis following myocardial infarction in rat. Am J Chin Med. 2007;35(2):279-95. Schulz F, Just I, Genth H. Prevention of Clostridium sordellii lethal toxin-induced apoptotic cell death by tauroursodeoxycholic acid. Biochemistry. 2009 Sep 29;48(38):9002-10.TalkVietnam. Bear bile tourism thrives despite exposés. April 5, 2012 at 12:30 pm. < http://talkvietnam.com/2012/04/bear-bile-tourism-thrives-despite-exposes/ > retrieved July 27, 2012Yoshizaki T, Kusunoki C, Kondo M, Yasuda M, Kume S, Morino K, Sekine O, Ugi S, Uzu T, Nishio Y, Kashiwagi A, Maegawa H. Autophagy regulates inflammation in adipocytes. Biochem Biophys Res Commun. 2012 Jan 6;417(1):352-7. Zhang T, Baehr W, Fu Y. Chemical Chaperone TUDCA Preserves Cone Photoreceptors in a Mouse Model of Leber Congenital Amaurosis. Invest Ophthalmol Vis Sci. 2012a Jun 5;53(7):3349-56.Zhang JY, Diao YF, Kim HR, Jin DI. Inhibition of endoplasmic reticulum stress improves mouse embryo development. PLoS One. 2012b;7(7):e40433. Epub 2012 Jul 13. Zhou L, Liu M, Zhang J, Chen H, Dong LQ, Liu F. DsbA-L alleviates endoplasmic reticulum stress-induced adiponectin downregulation. Diabetes. 2010 Nov;59(11):2809-16
What is tauroursodeoxycholic acid aka TUDCA ?
Tauroursodeoxycholic acid is a bile acid also known as TUDCA formed in the liver by conjugation of deoxycholate with taurine, usually as the sodium salt. While Western medicine has only gotten wind of the anti-apoptotic effects and its ability to protect mitochondria from cellular elements that would otherwise interfere with energy production within the past 20 years, or so, bear bile - you guessed it, a natural source of TUDCA - has an over 3,000 yearlong history of being used to treat visual disorders (Boatright. 2006), if which we know today that many of them can be treated and prevented by the administration of TUDCA and other bile-acids or bile-acid precursors, such as taurine (click here to read all about taurine)
So what's the suggested dose? That's actually quite a tricky question, because the number of human studies can be counted on the fingers of one hand (and this is still an exaggeration) and the available rodent data is mostly based on studies where the TUDCA was injected into the peritoneal cavity, which - depending on the compound that is used - will usually yield a bioavailabilty that's higher than what you will see with oral ingestion of the compound, yet lower than from intravenous injectionsAll of the following values are calculated for an 80kg human using the standard HED formula and are based on rodent studies showing benefits for the respective organ:Heart - 650mgDiabetes - 975mgLiver - 400-3,200mgAlzheimer's - 4,000mg*Parkinson's - 350mgPancreas - 3,250mg* This is the only value that's based on oral administration for the others I simply assumed 1:1 bioavailability which is probably already an overestimation for the liver and certainly for other tissues, since part of the TUDCA probably won't survive the first pass through the liver.
The distinction between different bile acids is difficile and beyond the scope of this mini-overview. What all of them have in common is that they are physiological detergents that facilitate excretion, absorption, and transport of fats and sterols in the intestine and liver. Bile acids are also steroidal amphipathic molecules derived from the catabolism of cholesterol, which makes them the ideal partner for both "water" and "fat soluble" molecules and allows them to do their job as major modulators of lipid secretion, bile flow and the absorption of dietary fats and vitamins. Aside from these long-established functions their role as regulators of key enzymes involved not only in cholesterol homeostasis, but also whole energy homeostasis has attracted more and more interest over the last years.
The unique detergent properties of bile acids are essential for the digestion and intestinal absorption of hydrophobic nutrients.No effect without "side effect"! Yet despite their metabolic utility bile acids have potent toxic properties and can - at high dosages - actually disrupt the very membranes they help to protext when they are administered at lower doses, therefore their accumulation in the blood and tissue is usually tightly regulated. And while Martinez-Diez et al. have shown that conjugated.bile acids have a very high toxicity threshold compared to their unconjugated cousins (Martinez-Diez. 2000), we are most probably still dealing with one of those classic U-shaped dose-response curves for TUDCA (with no effects at very low, beneficial effects at medium levels and toxicity issues at high levels) which are so ubiquitous in nature and still so difficult to understand for someone acculturated to the typical Western "more is more" mentality (Zinc would be another example, by the way; cf. "Zinc: 15mg is Plenty!").
A non-exhaustive list of proven benefitsWith its modulatory effects on the cell cylce regulator c-Jun N-terminal kinase (JNK), the generation and scavenging of radical oxigen specimen and glutathione S-transferase (GST) activity, which is necessary for the masteroxidant glutathione to do its job, TUDCA effectively prevented / reduced neurodegenation in rodent model of Parkinson's disease (Castro-Caldas. 2012). It has also been shown to reduce the amyloid beta-induced synaptic toxicity that is so characteristic of Alzheimer's disease (Nunes. 2012; Ramalho. 2012)Similar to taurine TUDCA appears to play an important role in cell osmolity, and has been shown to inhibit endoplasmic reticulum stress (ER) in embryonic development and ventricular contractile dsyfunction (malfunction of the heart) due to type II diabetes, fatty acid accumulation in the heart (Hua. 2010) and preserve cone density in the eye (Kim. 2012; Takada. 2012; Zhang. 2012a,b). The same anti-ER mechanism also protects the insulin releasing pancreatic beta cells of Wistar rats (Lee. 2010; Tang. 2012) and the kidney (Gao. 2012) from damage due to increased blood glucose. And Rivard et al. were even able to show that 400mg/kg of TUDCA, when they were administered intravenously were able to reduce apoptosis (death of hear cells) following myocardial infarction in rats (Rivard. 2007).TUDCA has also been shown to ameliorat insulin resistance in hypertrophic adipocytes (fat cells that burst from the seams; Jiao. 2011, Yoshizaki. 2012), to keep the expression of the adiponectine up (Zhou. 2010) and the inflammatory induced neovascularization in type II diabetes in check (Amin. 2012). Despite the fact that TUDCA is also effective against endoplasmic rectilium stress in the liver and in skeletal muscle it cannot save a methionine and choline deficient liver from getting clogged with fat (Henkel. 2012), or a palmitate (a saturated fatty acid) treated myotube from becoming insulin resistant, (Rieusse. 2012). What it can do, however is protect liver from apoptosis induced by natural or synthetic PPAR-gamma ligands (see "TTA + Fish Oil - Fat Burning Superfats or Hepatoxic Pro-Oxidants?" and "TTA + Fish Oil Revisited - Increased Intramuscular Omega-3 Levels Compromise Heart and Skeletal Muscle Performance"; cf. Nonaka. 2008) and ethanol-feeding (Colell. 2011)Interestingly the same anti-ER effects that protect the heart, the pancreas, the kidneys and, in the presence of adequate choline and methionine, also the liver, will also inhibit influenza A viral replication (Hassan. 2012; in-vitro data)TUDCA has also been found to have antibacterial effects, or rather to prevent the cytotoxic effects of Clostridium sordellii lethal toxin (CSLT) from virulent strains of Clostridium sordellii (Schulz. 2009) Only when it's conjugated to taurine UDCA (then T + UDCA = TUDCA ;-) will promote hydrocholeresis or, put simply, the output of bile acid (Úriz. 2011; intravenous administration). In view of the emerging importance of bile acids in overall energy expenditure and thyroid function (Ockenga. 2012).
This observation could well-explain the effects TUDCA has on the conversion of T4 => T3, which spiked your interest in the last installment of "On Short Notice", here at the SuppVersity (see "On Short Notice July 21, 2012").In this context it may also be wort mentioning that Nathanson et al. observed a direct stimulative effect of UDCA, the taurine devoid cousin of TUDCA on hepatic ATP secretion (Nathanson. 2001) and Drack et al. report that subcutaneous injections of 500 mg/kg in 0.15 M NaHCO3 to obesity prone mice reduced their weight gain by -22%; a significant effects on "normal" mice could was yet not observed in the study (Drack. 2012). And what about humans? As mentioned earlier, the number of studies in which TUDCA was actually administered to human beings is negligible and of actual relevance in the current context are probably on these two:Improved liver & muscle, but not adipose tissue insulin sensitivity in obese men and women (Kars. 2010) The main finding of this randomized double-blind study in which 20 obese subjects ([means +/- SD] aged 48 +/- 11 years, BMI 37 kg/m²) were assigned to receive either TUDCA at a dose of 1,750 mg/day or a placebo was a highly significant increase in insulin sensitivity (~30%; p < 0.05), which was - and this is would actually not be a bad thing for a physical culturist - liver and muscle specific!In combination with it's effect on the muscular and hepatic expression of p-AKT, this could make TUDCA the nutrient repartitioner (R-)ALA is not (cf. "Lean & Muscular with Alpha Lipoic Acid?"), if those effects would translate to individuals with normal insulin sensitivity (they do translate into human muscle cells in the Petri dish, which are protected against glucosamine induced decreases in GLUT-4 activity, when enough TUDCA was present in the incubation media; cf. Raciti. 2010).Surprisingly ineffective adjuvant after liver transplantation (Angelico. 1999) While you would expect that something that is good for liver health would also help as an adjuvant to the treatment of patients who received a liver transplant, the 16 subjects who were randomized to the active 2x250mg TUDCA treatment arm of the study did not see any statistically significant benefit in terms of one-year actuarial survival.Although the latter of these studies may actually be somewhat disappointing, its results are actually quite educative - after all, they put another emphasis on something I have touched upon in numerous previous blogposts, already: Even in cases as the one at hand, where the presence of statistically reduced serum cholesterol levels (p < 0.02) and less exuberant cholestasis confirm that a certain compound, in this case TUDCA, worked in vivo similarly to what it did in vitro, this does by no means implicate that it will also yield the real world results you would expect.
Figure 1: Anti-inflammatory effect of TUDCA on FFA treated adipocytes; reduction in TNF-alpha and IL-6 (also vs. baseline; directly from Jia. 2011))
Figure 2: Western blot analysis of influenza NP, NS1, and M1 proteins with corresponding densitometry displayed as averages with S.E. Proteins - reduced expression indicate a reduced replication rate (adapted from Hassan. 2011)
Figure 3: Effect of placebo or TUDCA on skeletal muscle insulin receptor substrate (IRS), Akt and JNK levels (Kars. 2010)
Implications: The "negative" or rather NULL outcome of the study by Angelico et al. should have reminded you that you better display a healthy degree of skepticism towards all the previously presented "scientifically established" benefits of TUDCA. Neither I, nor any of the researchers (not even Kars et al.) should have the hubris to tell you that taking X amounts of tauroursodeoxycholic acid will save your liver from the toxic effects of oral anabolic steroids, will help you in your battle against diabesity or make a powerful ally in your quest for a leaner, more muscular physique. And still, this mini-review of the literature should have shown you that TUDCA has a lot of potential, future studies will yet have to confirm, for whom these potentials can actually be realized and what dosages of a hitherto pretty pricey supplement will be necessary to produce the desired results... Needless to say that the SuppVersity is going to be the place wheer you will read about these studies first- right?
References: Amin A, Choi SK, Galan M, Kassan M, Partyka M, Kadowitz P, Henrion D, Trebak M, Belmadani S, Matrougui K. Chronic inhibition of endoplasmic reticulum stress and inflammation prevents ischaemia-induced vascular pathology in type II diabetic mice. J Pathol. 2012 Jun;227(2):165-74. Boatright JH, Moring AG, McElroy C, Phillips MJ, Do VT, Chang B, Hawes NL, Boyd AP, Sidney SS, Stewart RE, Minear SC, Chaudhury R, Ciavatta VT, Rodrigues CM, Steer CJ, Nickerson JM, Pardue MT. Tool from ancient pharmacopoeia prevents vision loss. Mol Vis. 2006 Dec 29;12:1706-14. Burroughs AK, Westaby D: Liver, biliary tract and pancreatic disease in Clinical Medicine, Eds. Kumar P, Clark M. Elsevier 2002Castro-Caldas M, Carvalho AN, Rodrigues E, Henderson CJ, Wolf CR, Rodrigues CM, Gama MJ. Tauroursodeoxycholic Acid Prevents MPTP-Induced Dopaminergic Cell Death in a Mouse Model of Parkinson's Disease. Mol Neurobiol. 2012 Jul 8.Chen Y, Liu CP, Xu KF, Mao XD, Lu YB, Fang L, Yang JW, Liu C. Effect of taurine-conjugated ursodeoxycholic acid on endoplasmic reticulum stress and apoptosis induced by advanced glycation end products in cultured mouse podocytes. Am J Nephrol. 2008;28(6):1014-22. Epub 2008 Jul 23.Colell A, Coll O, García-Ruiz C, París R, Tiribelli C, Kaplowitz N, Fernández-Checa JC. Tauroursodeoxycholic acid protects hepatocytes from ethanol-fed rats against tumor necrosis factor-induced cell death by replenishing mitochondrial glutathione. Hepatology. 2001 Nov;34(5):964-71.Drack AV, Dumitrescu AV, Bhattarai S, Gratie D, Stone EM, Mullins R, Sheffield VC. TUDCA slows retinal degeneration in two different mouse models of retinitis pigmentosa and prevents obesity in Bardet-Biedl syndrome type 1 mice. Invest Ophthalmol Vis Sci. 2012 Jan 5;53(1):100-6.Gao X, Fu L, Xiao M, Xu C, Sun L, Zhang T, Zheng F, Mei C. The nephroprotective effect of tauroursodeoxycholic Acid on ischaemia/reperfusion-induced acute kidney injury by inhibiting endoplasmic reticulum stress. Basic Clin Pharmacol Toxicol. 2012 Jul;111(1):14-23.Hassan IH, Zhang MS, Powers LS, Shao JQ, Baltrusaitis J, Rutkowski DT, Legge K, Monick MM. Influenza A viral replication is blocked by inhibition of the inositol-requiring enzyme 1 (IRE1) stress pathway. J Biol Chem. 2012 Feb 10;287(7):4679-89. Epub 2011 Dec 22.Henkel AS, Dewey AM, Anderson KA, Olivares S, Green RM. Reducing endoplasmic reticulum stress does not improve steatohepatitis in mice fed a methionine- and choline-deficient diet. Am J Physiol Gastrointest Liver Physiol. 2012 Jul;303(1):G54-9. Hua Y, Kandadi MR, Zhu M, Ren J, Sreejayan N. Tauroursodeoxycholic acid attenuates lipid accumulation in endoplasmic reticulum-stressed macrophages. J Cardiovasc Pharmacol. 2010 Jan;55(1):49-55. Jiao P, Ma J, Feng B, Zhang H, Diehl JA, Chin YE, Yan W, Xu H. FFA-induced adipocyte inflammation and insulin resistance: involvement of ER stress and IKKβ pathways. Obesity (Silver Spring). 2011 Mar;19(3):483-91. Lee YY, Hong SH, Lee YJ, Chung SS, Jung HS, Park SG, Park KS. Tauroursodeoxycholate (TUDCA), chemical chaperone, enhances function of islets by reducing ER stress. Biochem Biophys Res Commun. 2010 Jul 9;397(4):735-9.Kars M, Yang L, Gregor MF, Mohammed BS, Pietka TA, Finck BN, Patterson BW, Horton JD, Mittendorfer B, Hotamisligil GS, Klein S. Tauroursodeoxycholic Acid may improve liver and muscle but not adipose tissue insulin sensitivity in obese men and women. Diabetes. 2010 Aug;59(8):1899-905.Kim JS, Song BS, Lee KS, Kim DH, Kim SU, Choo YK, Chang KT, Koo DB. Tauroursodeoxycholic Acid Enhances the Pre-Implantation Embryo Development by Reducing Apoptosis in Pigs. Reprod Domest Anim. 2011 Dec 13.Martinez-Diez MC, Serrano MA, Monte MJ, Marin JJ. Comparison of the effects of bile acids on cell viability and DNA synthesis by rat hepatocytes in primary culture. Biochim Biophys Acta. 2000 Feb 21;1500(2):153-60.Nathanson MH, Burgstahler AD, Masyuk A, Larusso NF. Stimulation of ATP secretion in the liver by therapeutic bile acids. Biochem J. 2001 Aug 15;358(Pt 1):1-5.Nonaka M, Tazuma S, Hyogo H, Kanno K, Chayama K. Cytoprotective effect of tauroursodeoxycholate on hepatocyte apoptosis induced by peroxisome proliferator-activated receptor gamma ligand. J Gastroenterol Hepatol. 2008 Jul;23(7 Pt 2):e198-206.Nunes AF, Amaral JD, Lo AC, Fonseca MB, Viana RJ, Callaerts-Vegh Z, D'Hooge R, Rodrigues CM. TUDCA, a Bile Acid, Attenuates Amyloid Precursor Protein Processing and Amyloid-β Deposition in APP/PS1 Mice. Mol Neurobiol. 2012 Jun;45(3):440-54. Ockenga J, Valentini L, Schuetz T, Wohlgemuth F, Glaeser S, Omar A, Kasim E, duPlessis D, Featherstone K, Davis JR, Tietge UJ, Kroencke T, Biebermann H, Köhrle J, Brabant G. Plasma bile acids are associated with energy expenditure and thyroid function in humans. J Clin Endocrinol Metab. 2012 Feb;97(2):535-42.Ramalho RM, Nunes AF, Dias RB, Amaral JD, Lo AC, D'Hooge R, Sebastião AM, Rodrigues CM. Tauroursodeoxycholic acid suppresses amyloid β-induced synaptic toxicity in vitro and in APP/PS1 mice. Neurobiol Aging. 2012 May 21.Rieusset J, Chauvin MA, Durand A, Bravard A, Laugerette F, Michalski MC, Vidal H. Reduction of endoplasmic reticulum stress using chemical chaperones or Grp78 overexpression does not protect muscle cells from palmitate-induced insulin resistance. Biochem Biophys Res Commun. 2012 Jan 6;417(1):439-45. Sokol RJ, Dahl R, Devereaux MW, Yerushalmi B, Kobak GE, Gumpricht E. Human hepatic mitochondria generate reactive oxygen species and undergo the permeability transition in response to hydrophobic bile acids. J Pediatr Gastroenterol Nutr. 2005 Aug;41(2):235-43.Takada A, Miki T, Kuno A, Kouzu H, Sunaga D, Itoh T, Tanno M, Yano T, Sato T, Ishikawa S, Miura T. Role of ER Stress in Ventricular Contractile Dysfunction in Type 2 Diabetes. PLoS One. 2012;7(6):e39893. Epub 2012 Jun 29.Tang C, Koulajian K, Schuiki I, Zhang L, Desai T, Ivovic A, Wang P, Robson-Doucette C, Wheeler MB, Minassian B, Volchuk A, Giacca A. Glucose-induced beta cell dysfunction in vivo in rats: link between oxidative stress and endoplasmic reticulum stress. Diabetologia. 2012 May;55(5):1366-79.Úriz M, Sáez E, Prieto J, Medina JF, Banales JM. Ursodeoxycholic acid is conjugated with taurine to promote secretin-stimulated biliary hydrocholeresis in the normal rat. PLoS One. 2011;6(12):e28717. Epub 2011 Dec 14. Raciti GA, Iadicicco C, Ulianich L, Vind BF, Gaster M, Andreozzi F, Longo M, Teperino R, Ungaro P, Di Jeso B, Formisano P, Beguinot F, Miele C. Glucosamine-induced endoplasmic reticulum stress affects GLUT4 expression via activating transcription factor 6 in rat and human skeletal muscle cells.Diabetologia. 2010 May;53(5):955-65. Rivard AL, Steer CJ, Kren BT, Rodrigues CM, Castro RE, Bianco RW, Low WC. Administration of tauroursodeoxycholic acid (TUDCA) reduces apoptosis following myocardial infarction in rat. Am J Chin Med. 2007;35(2):279-95. Schulz F, Just I, Genth H. Prevention of Clostridium sordellii lethal toxin-induced apoptotic cell death by tauroursodeoxycholic acid. Biochemistry. 2009 Sep 29;48(38):9002-10.TalkVietnam. Bear bile tourism thrives despite exposés. April 5, 2012 at 12:30 pm. < http://talkvietnam.com/2012/04/bear-bile-tourism-thrives-despite-exposes/ > retrieved July 27, 2012Yoshizaki T, Kusunoki C, Kondo M, Yasuda M, Kume S, Morino K, Sekine O, Ugi S, Uzu T, Nishio Y, Kashiwagi A, Maegawa H. Autophagy regulates inflammation in adipocytes. Biochem Biophys Res Commun. 2012 Jan 6;417(1):352-7. Zhang T, Baehr W, Fu Y. Chemical Chaperone TUDCA Preserves Cone Photoreceptors in a Mouse Model of Leber Congenital Amaurosis. Invest Ophthalmol Vis Sci. 2012a Jun 5;53(7):3349-56.Zhang JY, Diao YF, Kim HR, Jin DI. Inhibition of endoplasmic reticulum stress improves mouse embryo development. PLoS One. 2012b;7(7):e40433. Epub 2012 Jul 13. Zhou L, Liu M, Zhang J, Chen H, Dong LQ, Liu F. DsbA-L alleviates endoplasmic reticulum stress-induced adiponectin downregulation. Diabetes. 2010 Nov;59(11):2809-16
Attachments
Last edited: